14 Nov Reunion Neuroscience Inc. Provides Business Update and Reports Fiscal Second Quarter 2023 Financial Results
- Completed RE104 Phase 1 prespecified dose cohorts and on track to deliver interim Phase 1 analysis in the first calendar quarter of 2023.
- Appointed Greg Mayes as President and Chief Executive Officer, with Joseph del Moral moving into the role of Chairman.
- Announced reinforcement of the executive leadership team with the addition of highly experienced pharmaceutical professionals: Edward Smith as Chief Financial Officer, Dr. Aviva Asnis-Alibozek as Vice President, Medical Affairs, and Curtis Weber as General Counsel.
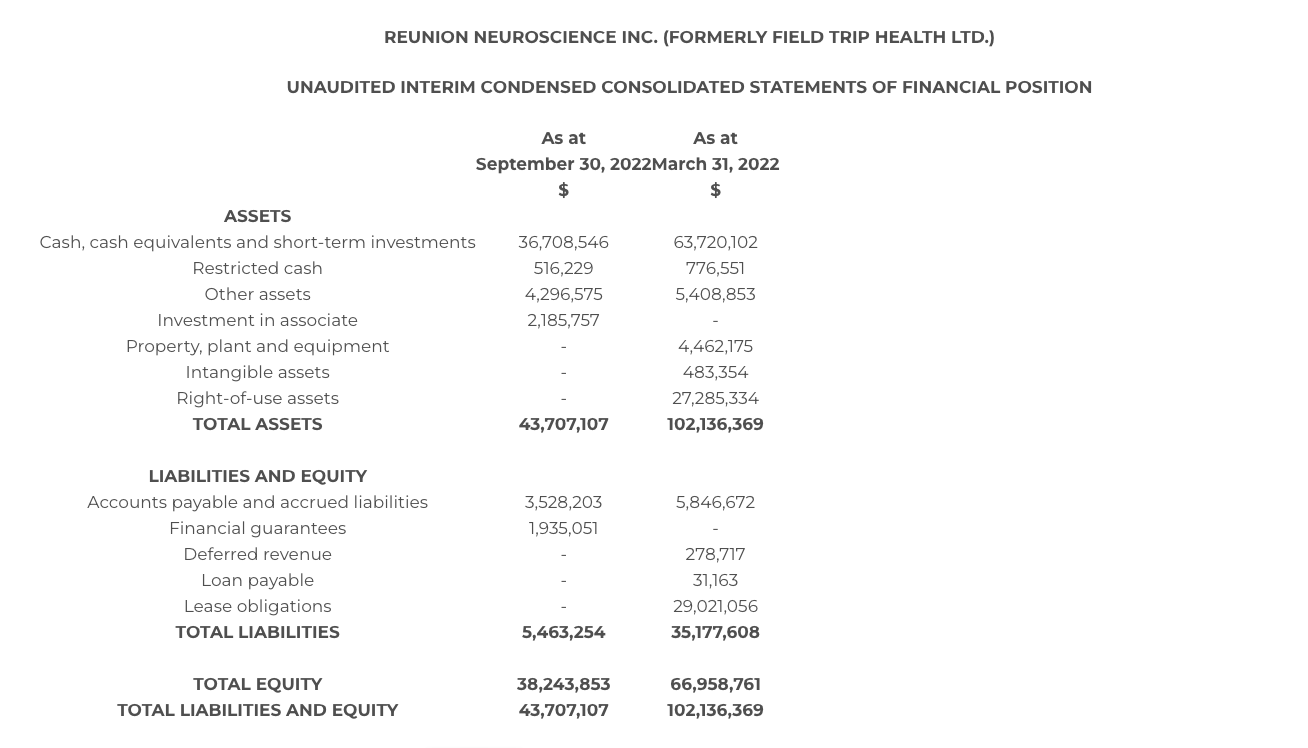
- At September 30, 2022, the Company held cash, cash equivalents and short-term investments of $36.7 million Canadian dollars.
TORONTO, Nov. 14, 2022 (GLOBE NEWSWIRE) — Reunion Neuroscience Inc. (NASDAQ: REUN, TSX: REUN), a biopharmaceutical company committed to developing innovative and patented therapeutic solutions for underserved mental health conditions, today reported its business update and fiscal second quarter ended September 30, 2022 results.
Greg Mayes, President & Chief Executive Officer of Reunion, said, “Reunion has hit the ground running as an independent company, advancing our mission of developing innovative and patent protected therapeutic solutions for mental health conditions for the hundreds of millions of people with unmet mental health needs. We are well positioned to round out the year in preparation for what promises to be an exciting 2023. I am thrilled to announce that we have dosed all subjects in the first four cohorts of our Phase 1 study with a planned readout set for Q1 2023 and have strengthened our leadership team, appointing seasoned industry professionals in the roles of Chief Financial Officer, Vice President of Medical Affairs, and General Counsel to support our next stage of growth.”
Key Highlights and Recent Developments
RE104
Reunion is developing its lead clinical-stage drug candidate, RE104, a patent protected novel serotonergic psychedelic designed to deliver quick onset, limited duration psychoactivity and durable antidepressant activity. RE104, the only novel prodrug of 4-OH-DIPT, aims to reduce the treatment burden for patients and payers and provide a safe and effective new therapeutic option for the large population of underserved patients suffering from postpartum depression, and potentially other mental health indications.
Reunion announced the first successful dosings in the RE104 Phase 1 Clinical Study in July 2022, and subsequent to quarter end, completed the dosing of the four prespecified cohorts, representing 32 subjects, for its planned interim analysis to be released in the first calendar quarter of 2023. The study aims to determine the safety, tolerability, pharmacokinetics and pharmacodynamic effects of RE104 over a range of dosage levels. Results of subjective measurements including intensity, duration and feelings produced by the period of acute psychoactivity after administration of RE104 will be used to select an appropriate recommended phase 2 dose (RP2D). Per the Phase 1 protocol, up to two additional dosing cohorts may be evaluated based on pharmacokinetics, pharmacodynamics and safety.
Data generated from the interim analysis will be used to support its planned pre-IND regulatory interactions with the United States Food and Drug Administration (US FDA) in early 2023 where the Company will also present plans for opening an Investigational New Drug (IND) application later in the year for the development of RE104 as a treatment for postpartum depression.
Dr. Nathan Bryson, Chief Scientific Officer, Reunion, commented, “The rapid enrollment of our RE104 Phase 1 clinical study clearly demonstrates significant progress toward our goal of advancing a differentiated molecule which could offer a convenient and effective therapeutic option for patients who suffer from depression and other mental health disorders. We look forward to sharing data from the Phase 1 study in the first quarter of 2023 and laying the groundwork in preparation for our RE104 Phase 2 program.”
RE200 Series
Reunion is also developing the RE200 series. These novel molecules are structurally similar to classical psychedelics, but have selective potency at the target serotonin 2A receptor (5HT2A) and are devoid of 5HT2B receptor agonism. During the second quarter, Reunion progressed in its understanding of some of the structural features that define this class of new chemical entity (NCE) molecules, initiated preclinical work to identify potential drug candidates in the RE200 series and strengthened its patent portfolio. The Company plans to nominate its first lead clinical candidate from the RE200 series in 2023.
Corporate Updates
In September, the Company appointed Greg Mayes as President and Chief Executive Officer, with Joseph del Moral stepping into the role of Chairman. Mr. Mayes’ initial focus is on the successful completion of the Company’s Phase 1 clinical trial with RE104 and the planned launch of its Phase 2 study in postpartum depression. Mr. Mayes will also progress Reunion’s preclinical RE200 research focused on the discovery of selective and specific serotonergic psychedelics.
Post quarter end, Reunion announced the appointment of Edward Smith as the Company’s Chief Financial Officer, Dr. Aviva Asnis-Alibozek as Vice President, Medical Affairs and Curtis Weber as General Counsel.
Mr. Mayes continued, “Reunion’s strong composition of matter intellectual property significantly differentiates us from other psychedelic therapeutic companies. Making Reunion even more exciting is the Company’s development program, which targets a niche initial indication with high unmet therapeutic need – PPD. We firmly believe that our IP is what distinguishes us, our choice of molecule differentiates us, and our team is what will separate us from the growing number of companies in this space. These factors will continue to generate further interest in Reunion as we seek to build partnerships and expand our clinical pipeline toward the future.”
Corporate Structure and Basis of Presentation
On August 11, 2022, the Company completed its previously announced spinout of its clinics and botanical research operations (Clinic Operations) to Field Trip Health & Wellness with the resulting drug discovery and development business renamed Reunion Neuroscience Inc., which is listed on the NASDAQ Stock Market and Toronto Stock Exchange under the ticker symbol “REUN”.
Reunion accounted for the Clinic Operations as discontinued operations whose assets and liabilities are classified and presented separately as current items held for transfer in the statement of financial position and are measured at their carrying amount. Clinic Operations are excluded from the results of continuing operations and are presented as a single amount as a net loss from discontinued Clinic Operations in the unaudited interim condensed consolidated statements of loss.
For more details on the spinout transaction, please refer to the Key Highlights and Recent Developments – Reorganization and Spinout of Clinic Operations section of the Company’s management’s discussion and analysis, available under the Company’s SEDAR profile at www.sedar.com.
Financial Highlights
Selected Condensed Consolidated Financial Information
The following table sets forth selected condensed consolidated financial information derived from the Company’s unaudited consolidated financial statements for the fiscal second quarter 2023 ended September 30, 2022, prepared in accordance with IAS 34 in a manner consistent with the Company’s annual audited financial statements, which are reported under International Financial Reporting Standards and in Canadian dollars. The following information should be read in conjunction with the financial statements and management’s discussion and analysis, which are available on the Company’s website at www.reunionneuro.com and under the Company’s SEDAR profile at www.sedar.com.
Overview of operations
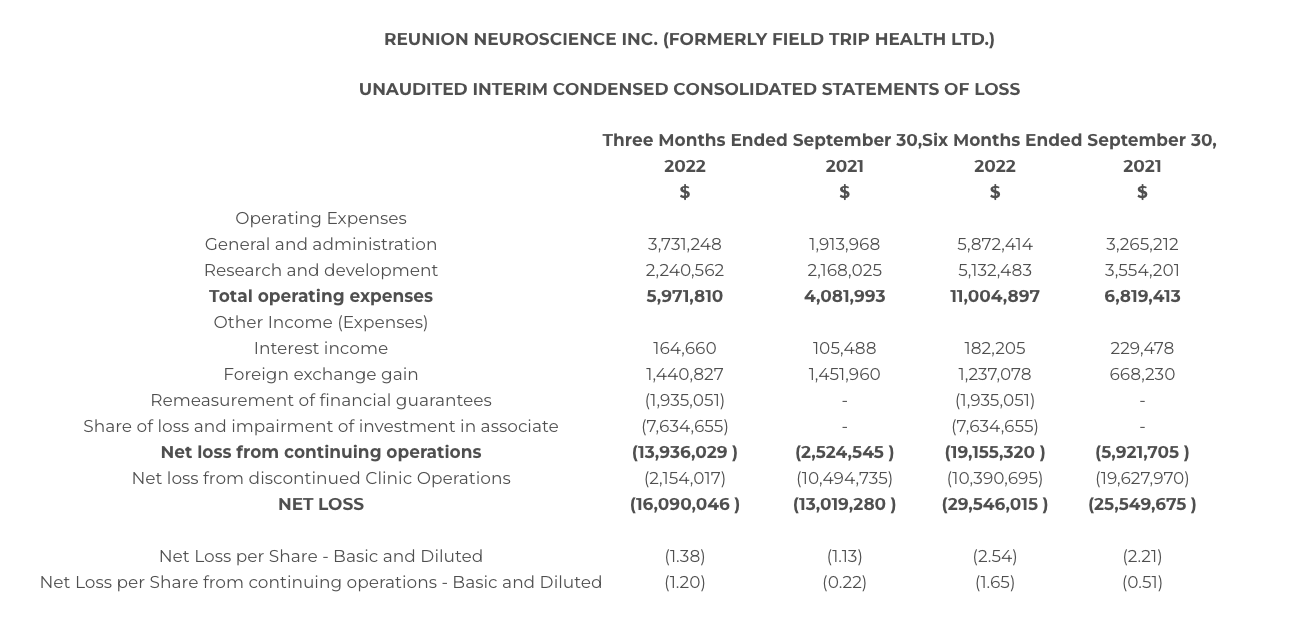
The Company incurred general and administrative expenses of $3.7 and $5.9 million for the three and six month periods ended September 30, 2022 compared to $1.9 and $3.3 million for the same periods in 2021. Increases were attributable to increased headcount and other costs associated with the becoming a public Company with increased scale of operations due to the Company entering the clinical stage for its lead asset RE104.
Research and development expenses of $2.2 and $5.1 million were incurred for the three and six month periods ended September 30, 2022 compared to $2.2 and $3.6 million for the same periods in 2021. Increases were attributable to personnel and third-party manufacturing and clinical research costs associated with the ongoing Phase 1 clinical trial for RE104.
Other income and expenses include interest income on the Company’s cash, cash equivalents and investment balances and foreign currency gains primarily attributable to the Company’s United States dollar holdings. The Company also recognized $1.9 and $7.6 million in non-cash charges for the three and six months ended September 30, 2022 in recognition of (i) its financial guarantee of certain lease obligations associated with entities that were part of the spinout of Clinic Operations and (ii) the Company’s equity share of loss and impairment of its investment in Field Trip Health & Wellness, respectively.
The Company recognized a net loss from discontinued operations of $2.2 and $10.4 million for the three and six month periods ended September 30, 2022 compared to $10.5 and $19.6 million for the same periods in 2021. Discontinued operations are attributable to the spinout of Clinic Operations completed on August 11, 2022.
The Company incurred a net loss from continuing operations of $13.9 million or $1.20 per share and $19.2 million or $1.65 per share for the three and six month periods ended September 30, 2022 compared to a loss of $2.5 million or $0.22 per share and $5.9 million or $0.51 per share for the three and six month periods ended September 30, 2021.
At September 30, 2022, the Company held cash, cash equivalents and investments of $36.7 million.
Conference Call
The Company will conduct a conference call and webcast to discuss its results on Monday, November 14, 2022, at 8:30 a.m. ET. To access the call, please dial 1-877-407-9716 (within the U.S.) or 1-201-493-6779 (outside the U.S.) and provide conference ID 13734045. A live webcast of the conference call can be accessed via the Events and Presentations section of the Reunion Investor Relations website here.
For those unable to attend the live call, a telephonic replay will be available until 11:59 p.m. ET on Monday, November 21, 2022. To access the replay dial 1-844-512-2921 (within the U.S.) or 1-412-317-6671 (outside the U.S.) and provide conference ID 13734045. The webcast will be archived and available in the Events and Presentations section of the Reunion Investor Relations website approximately one hour after the conclusion of the live call.
About Reunion Neuroscience Inc.
Reunion (formerly, Field Trip Health Ltd.) is committed to developing innovative therapeutic solutions for mental health conditions. The Company’s lead asset, RE104, is a proprietary, novel serotonergic psychedelic compound being developed as a potential fast-acting and durable antidepressant for patients suffering from postpartum depression and other mental health conditions. The U.S. Patent and Trademark Office has granted the Company a patent for the claims related to RE104, granting it exclusive rights to the composition of matter, use and manufacturing of a family of hemi-ester compounds of hydroxytryptamines, including RE104. The patent will provide protection until 2041. Reunion is also developing the RE200 series, which includes compounds with potential for more selective serotonin receptor activity with reduced psychoactivity for potential use in more chronic treatment paradigms and indications.
Cautionary Note Regarding Forward-Looking Information
This release includes forward-looking information (within the meaning of Canadian securities laws and within the meaning of the United States Private Securities Litigation Reform Act of 1995) regarding Reunion and its business. Often but not always, forward-looking information can be identified by the use of words such as “expect”, “intends”, “anticipates”, “plans”, “believes” or variations (including negative variations) of such words and phrases, or state that certain actions, events or results “may”, “could”, “would”, “should” or “will” be taken, occur or be achieved. Such statements are based on the current expectations and views of future events of the management of Reunion and are based on assumptions and subject to risks and uncertainties, many of which are beyond Reunion’s control. Although the management of Reunion believes that the assumptions underlying these statements are reasonable, they may prove to be incorrect. The forward-looking events and circumstances discussed in this release may not occur and could differ materially as a result of known and unknown risk factors and uncertainties affecting the companies, including the funds available to Reunion and the use of such funds, the timing, completion and potential outcome of testing and research on Reunion’s drug trial candidates, RE104 and the RE 200 Series, including the ability to recruit patients, to retain and identify clinical partners, and to optimize dosage amounts, the likelihood and ability of Reunion to complete an investigational new drug application and obtain regulatory approvals, as required, prior to initiating further clinical trials for RE104 and molecules within the RE200 Series, the ability of Reunion to meet eligibility requirements for clinical testing and through to more complex clinical trials, the ability of Reunion to protect and expand its intellectual property portfolio, the performance of Reunion’s affiliate, Field Trip Health & Wellness Ltd., the ability of Reunion to produce and supply its drug trial candidates, market conditions, economic factors, management’s ability to manage and to operate the business, the equity markets generally and this and other Risk Factors disclosed in Reunion’s public filings available on the SEDAR website at www.sedar.com and on the EDGAR section of the SEC’s website at www.sec.gov. Although Reunion has attempted to identify important factors that could cause actual actions, events or results to differ materially from those described in forward-looking statements, there may be other factors that cause actions, events or results to differ from those anticipated, estimated or intended. Accordingly, readers should not place undue reliance on any forward-looking statements or information. No forward-looking statement can be guaranteed. Except as required by applicable securities laws, forward-looking statements speak only as of the date on which they are made (or such earlier date, if identified) and Reunion does not undertake any obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise. Additional information relating to Reunion, including its Annual Information Form and Risk Factors, can be located on the SEDAR website at www.sedar.com and on the EDGAR section of the SEC’s website at www.sec.gov.
This press release does not constitute an offer to sell or the solicitation of an offer to buy securities.
Neither the Toronto Stock Exchange, nor its Regulation Services Provider, have approved the contents of this release or accept responsibility for the adequacy or accuracy of this release.
CONTACTS:
Reunion Neuroscience:
Greg Mayes
President & CEO
(215) 696-9659
gmayes@reunionneuro.com
Investor contacts:
Phil Carlson / Sophia Bashford
KCSA Strategic Communications
(646) 573-0776 / (929) 246-7307
reunion@kcsa.com
Media contacts:
Shana Marino
KCSA Strategic Communications
(347) 487-6189
reunion@kcsa.com
SOURCE Reunion Neuroscience Inc.